Environmental Chemistry Project
The Retention of Harmful Metals by Biochar in Mine Drainage Soils
Hannah Quick, Stephan Davoust, Cooper Stowers
In affiliation with Animas High School
Abstract
While there have been several studies previously conducted using the incorporation of biochar at a high altitude with the intent of remediating soil and plant growth affected by acid mine drainage in the environment, the effective retention rate of harmful metals (such as lead, iron, copper, manganese, aluminum, and zinc) in soil leachate has not yet been correlated to a specific concentration of biochar. Biochar was added to soil samples that had been taken from five different remediation areas to in order to conduct several small-scale trials conducted in a lab. Biochar was shown to reduce metal levels at certain concentrations and was shown to cause significant plant and vegetation growth once the samples had been collected, but there was no uniform concentration at significant decreases when exposed to a 10% biochar concentration, but just as many other metals showed much more erratic results, making the correlations between the changes in metals and a specific concentration of biochar undeterminable. We conclude that that biochar addition to soil does help lower levels of harmful metals, but in an unpredictable manner.
1. Introduction
The contamination of soil in high altitude mine drainage sites plays a determining role in the ecological and environmental characteristics in the areas surrounding these sites. One of the key factors that verifies the harmful effects caused by acid mine drainage is the introduction of harmful metals into the environment either through water contamination or direct soil contact (Beesley and Marmiroli 2010). Acid mine drainage is a natural consequence of mining activity where the excavation of mineral deposits (metal bearing or coal), below the natural groundwater level, exposes inorganic compounds to oxygen and water (McGuinness, S 1999). Environmentally hazardous metals such as Cu, Pb and Cd can be considered harmful to organic and inorganic materials. Direct effects of acid mine drainage include erosion of naturally formed river beds and manmade structures, birth defects and in severe cases death of aquatic life, termination of submerged and exposed vegetation, and rising levels in pH of waters in downriver rivers and other bodies of water (Earle and Callaghan 2012). Previously published research shows that biochar, a low-density pyrolyzing material produced by burning biomass under conditions of low temperature and minimal oxygen (Beesley and Marmiroli 2010),” neutralizes the toxic qualities of organic and inorganic pollutants. Biochar may be added to soils with the intention to improve soil functions and to reduce emissions from biomass that would otherwise naturally degrade to greenhouse gases (International Biochar Archive). In order to test this, soil samples were collected at the Joe Johnson, Bonner, Across from Bonner, Brooklyn, and Road Cut mining sites, where biochar had not been introduced to the soil. The mine site has been linked as a key factor to the damages inflicted by acid mine drainage. After the samples were transported to a private research facility, each sample was processed using various metal analysis equipment to provide accurate metal concentration percentages and monitor the effectiveness of Biochar in given percentages corresponding with designated mine sites. Biochar has been proven to retain specific metals (Mendez and Maier 2008) as a means of lowering the direct influence of negative environmental effects thus contributing to the remediation of acid mine drainage sites (Uchimiya, Chang and Klasson 2011). This draws the hypotheses that the biochar will have a high metal retention percentage and allow for sufficient rehabilitation of environments affected by acid mine drainage.
Table 1: The GPS coordinates of the different mine sites that were tested
2. Methods & Materials
Field scientists from Mountain Studies Institute based in Silverton, Colorado, gathered at the Joe John, Brooklyn, Bonner, Across from Bonner, and Road Cut mine sites in Silverton in September of 2011 to collect soil samples. Soil samples were taken back to the facility in order to collect water samples weekly, and take leachate samples at the beginning and the end of experiment was a greenhouse/container experiment where we measured soil water content, soil water pH, and plant growth response. The trial was comprised of concurrent trials where student at Animas High School grew an alpine restoration seed mix in 60 3.5-inch containers under grow lights for 55 days.
2.1 Sample Preparation
When collecting samples, scientists collected one five-gallon bucket-full sample of soil from each of the five mine sites. The soil was then transported to Animas High School, where students prepared the samples in containers. Students first sieved the sample in order to separate out the soil from the rocks, as well as to break the particles down to less than 2 mm fractions. 200 ml of each soil sample were weighed in grams and recorded on the data sheet. Once the samples had been broken down, a mix of seeds was separated into units of one gram, in addition to 4 Lupine seeds. The seed mixture was added in a labeled container to the soil sample later on in the experiment. After the soil and seed mixtures had been sorted out, the biochar volumes were measured in accordance to the equalities: 0 ml=0%, 20ml=10%, 40ml=20%, and 60ml=30% of biochar per 200 ml soil sample. These volumes were then converted and recorded as mass measured in grams. Once all of the data was recorded, the soil and biochar were thoroughly mixed together in their respective containers. The samples were mixed together by combining the two in a mixing container as completely as possible, and then returning the samples to their labeled containers. Next, the mixture was saturated with water and left to drain overnight. Once drained, the biochar and soil were gently mixed back together, and the seed combination was added to the mixture. Once the seeds were added, the mixture was saturated, drained, and finally weighed. After the mass had been recorded, the sample was covered and left to germinate for 72 hours at a warm temperature. Once the seeds had germinated, the samples were watered once a week with 100 ml of tap water. The pH of the tap water was recorded before irrigation.
2.2 Measurements Taken Throughout Trial
After the samples had been correctly set up, measurements were taken once a week for the duration of the study. Students first documented the samples photographically, noting the site plot, treatment, date, and team on a dry erase board that was included in the photo. Students then recorded the average height (rounded to the nearest 5 mm) of the green vegetation that had grown in the sample. Students then irrigated the samples with 100 ml of tap water, and recorded the mass of the sample in grams. Mass of the sample was also taken prior to irrigation. Once the water had been drained, the pH of the leachate was recorded. For the first and last irrigations of the study, the leachate was collected for metals analysis.
2.3 Metals Analysis
In order to collect the leachate, the initial leachate samples were filtered with filter paper into 60 ml plastic bottles. Upon collection, they were refrigerated, in accordance with standard protocol. They were then fixed with 12 drops of 70% nitric acid by mass in order to keep the dissolved metals in their dissolved forms. Once the leachate had been collected, the samples were sent to a lab in Fort Collins, Colorado for metals analysis in an ICP-MS instrument. ICP-MS stands for Inductively Coupled Plasma Mass Spectrometry. The first step in the process is converting the sample to an aerosol in order to excite the particles and carry them into the plasma. The objective of the plasma is to strip the electrons from the analyte atoms and to generate ions. The ions are then transmitted through a spectrometer and filtered by their mass, thereby emitting a mass spectrum in correlation to the element of the particles. The elements are identified by associating the peak in the spectrum to the known isotope concentration of the element. (Thermo Fisher, 12-14).
3. Results
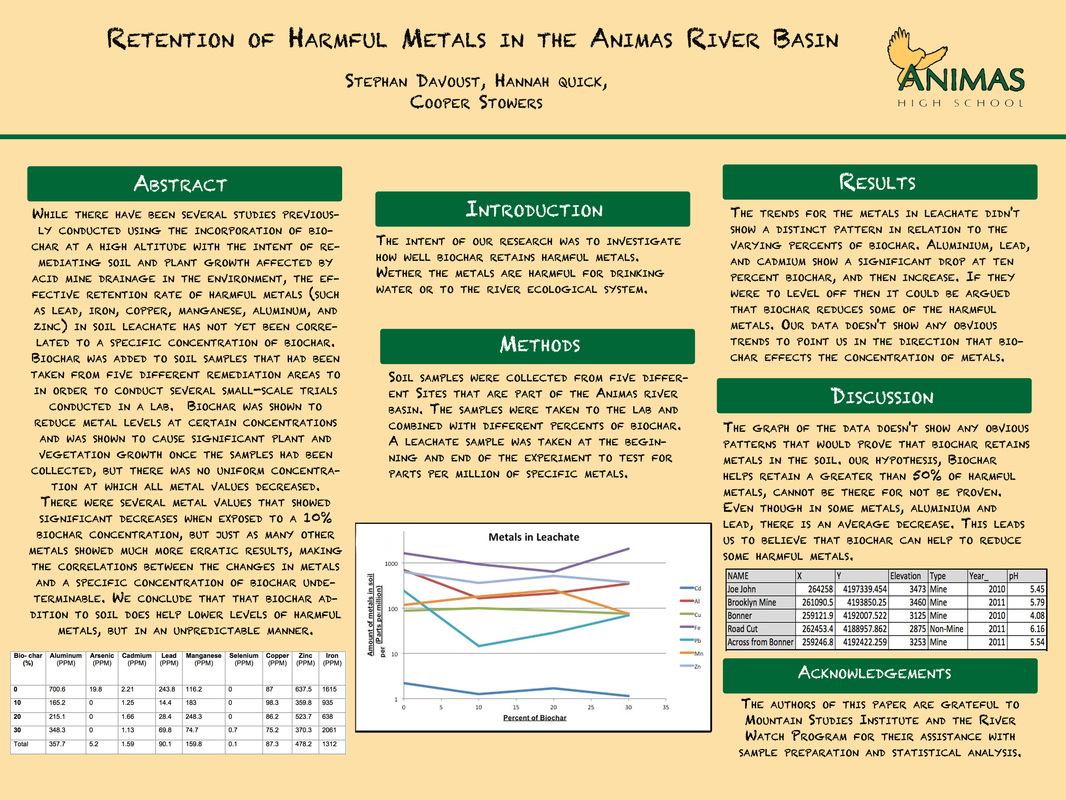
The results of distributing varying concentrations of biochar throughout different mine sites in Silverton, Colorado, suggest that biochar successfully retains harmful metals in solution, such as aluminum, cadmium, lead, copper, zinc, iron, arsenic, manganese, and selenium to a certain degree. Although some data remains inconsistent in terms of metal retention for some concentrations of biochar, specifically concentrations of biochar at 20% and 30%, the overall spectrum of the data shows that biochar can cause a significant reduction of metal concentration in soil.
Lead displayed a significant decrease in concentrations when exposed to 10% biochar concentration. After being exposed to higher concentrations, however, the lead concentrations began to increase at a rate that was slower than the initial decrease, but still consistent. aluminum showed a significant reduction in concentration when exposed to a 10% biochar concentration.
Iron presented high levels in the leachate, but once introduced to biochar, those levels started decreasing gradually. Once the biochar concentrations reached above 20%, however, levels of iron started spiking.
Because of the significant differences between biochar concentrations and their effectiveness in retaining both aluminum and iron, there is most likely no relationship between biochar concentrations and their abilities to retain iron or aluminum. Once the biochar was mixed with a 20% concentration of biochar, however, there was a slight decrease in levels of copper present in the leachate.
Manganese did not follow this trend. Instead of the amounts of metals decreasing after being introduced to biochar, the levels increased until they were exposed to 20% biochar concentrations, when the levels at last started to decrease.
The levels of Cadmium were so low that there was hardly any change when the sample was introduced to biochar. Nevertheless, levels of cadmium did slightly decrease after being mixed with concentrations of biochar.
Zinc simply showed erratic results, making it difficult to draw any substantial data from the results.
The average retention rate for all metals at 10% biochar concentration was 32.7%, 20% biochar concentration was 22.09%, and 30% biochar concentration averaging out to 35.9%. The median retention rates of all of these biochar concentrations was 30.23%, with the mode being 52%. The range between all of rates of retention represented a difference of 208, with levels ranging between -113% retention and 95%.
Figure 2: the overall retention rate of metals at the given percent biochar
4. Discussion
Because there were several obvious retentions in certain metal levels, there is a possibility that biochar is only effective in retaining certain metals at a certain concentration. For example, it is apparent in the graphs that biochar is somewhat successful in retaining aluminum and lead levels at concentrations equal to or less than 20%, because both metal values started spiking after 20% biochar concentrations. Iron levels are also reduced when exposed to anything less than a 20% biochar concentration. Contrary to these results, both manganese and copper started decreasing in values only when exposed to concentrations greater than 20%. Regardless of the biochar concentrations zinc was exposed to, there was no apparent reaction to the added substance, suggesting biochar does not help retain zinc in solution. Because of the variance in results, we have concluded that there is no specific biochar concentration that will be successful in retaining the harmful metals listed above. There is a slight possibility, however, that the inclusion of biochar in the soil sample raised the pH of the metals, thereby decreasing the potential of the metals in solution to sorb to the biochar, which is why we think that our results were so varied.
Because of the variance in our results, we cannot prove our hypothesis valid, as there was no uniform decrease in the metal values. This variance may be related to errors in measuring metal values, as there were some values that seemed very random or odd in relation to other measurements in the same study. These errors would have affected the results in a random way, either increasing or decreasing the estimated retention rate, as was obvious when the calculated average retention rate was greater than negative or greater than 100%. It is also not certain that the plants all got the same treatment, or the leachate was collected the same, which would result in randomized data once again. Because this is an original study in the respect that no other studies have tested varying concentrations of biochar, our results are not comparable to another published study. One study, A review of Biochars’ Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soils, did state in their abstract that “Specific mechanisms of contaminant-biochar retention and release over time and the environmental impact of biochar amendments on soil organisms remain somewhat unclear but must be investigated to ensure that the management of environmental pollution coincides with ecological sustainability,” stressing the importance of biochar as a combatant towards certain types of environmental pollution, including acid mine drainage. For future studies, it may prove beneficial to send the soil samples to two different metals analysis centers, in order to compare results and confirm the accuracy of the values.
5. Acknowledgements
The authors of this paper are grateful to Mountain Studies Institute and the River Watch Program for their assistance with sample preparation and statistical analysis.
6. References
Beesley, Luke, Eduardo Moreno-Jimenéz, Jose L. Gomez-Eyles, Eva Harris, Brett Robinson, and Tom Sizmur. A Review of Biochars ’ Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soils. Publication. A Review of Biochars' Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soilsand Restoration of Contaminated Soils (Tom Sizmur). El Sevier, 2011. Web. 15 Apr. 2012.
Beesley, L. and Marmiroli, M., 2010. “The immobilization and retention of soluble arsenic, cadmium and zinc by biochar.” Environmental Pollution pp. 1
Colorado Department of Public Health and Environment 2009. System Classifacation and Water Quality Standards.”Water Quality Regulations AnimasPDF
Colorado Department of Public Health and Environment 2009.“The Basic Standards for Ground Water.” The Water Quality Control CommissionPDF
Earle, J., Callaghan, T., “Impacts of mine drainage on aquatic life, water uses, and man-made structures.” in Coal Mine Drainage Prediction and Prevention Department of Environmental Protection, Harrisburg, PA 17105 pp. 4-1 - 4-10
“From First Principles: An Introduction to the ICP-MS Technique.” Thermo Fisher Scientific. Web. 18 Apr 2012.http://www.thermo.com/CMA/PDFs/Various/File_2512.pdf
Jonesw, DL., Rousk, J., Edwards-Jones, G., DeLuca,T.H., Murphy, D.V.Murphy, 2011 “Biochar-mediated changes in soil quality and plant growth in a three year trial.” Soil Biology and Biochemistry pp. 113-124
McGuinness, S., 1999. Treatment of acid mine drainage. RESEARCH PAPER 99/10 House of Commons pp. 1-5
Mendez, M. and Maier, R., 2008. “Phytostabilization of Mine Tailings in Arid and Semiarid Environments- An Emerging Remediation Technology.” Department of Soil, Water, and Environmental Science, University of Arizona, Tucson, Arizona, USA pp.278-280
Uchimiya, M., Chang,, S., Klasson, T., 2011“Screening biochars for heavy metal retention in soil: Role of oxygen functional groups” Journal of Hazardous Materials, Volume 190, Issues 1–3, 15 June 2011, pp. 432-441
"What Is Biochar?" International Biochar Initiative. 2012. Web. 02 May 2012. <http://www.biochar-international.org/biochar>.
Hannah Quick, Stephan Davoust, Cooper Stowers
In affiliation with Animas High School
Abstract
While there have been several studies previously conducted using the incorporation of biochar at a high altitude with the intent of remediating soil and plant growth affected by acid mine drainage in the environment, the effective retention rate of harmful metals (such as lead, iron, copper, manganese, aluminum, and zinc) in soil leachate has not yet been correlated to a specific concentration of biochar. Biochar was added to soil samples that had been taken from five different remediation areas to in order to conduct several small-scale trials conducted in a lab. Biochar was shown to reduce metal levels at certain concentrations and was shown to cause significant plant and vegetation growth once the samples had been collected, but there was no uniform concentration at significant decreases when exposed to a 10% biochar concentration, but just as many other metals showed much more erratic results, making the correlations between the changes in metals and a specific concentration of biochar undeterminable. We conclude that that biochar addition to soil does help lower levels of harmful metals, but in an unpredictable manner.
1. Introduction
The contamination of soil in high altitude mine drainage sites plays a determining role in the ecological and environmental characteristics in the areas surrounding these sites. One of the key factors that verifies the harmful effects caused by acid mine drainage is the introduction of harmful metals into the environment either through water contamination or direct soil contact (Beesley and Marmiroli 2010). Acid mine drainage is a natural consequence of mining activity where the excavation of mineral deposits (metal bearing or coal), below the natural groundwater level, exposes inorganic compounds to oxygen and water (McGuinness, S 1999). Environmentally hazardous metals such as Cu, Pb and Cd can be considered harmful to organic and inorganic materials. Direct effects of acid mine drainage include erosion of naturally formed river beds and manmade structures, birth defects and in severe cases death of aquatic life, termination of submerged and exposed vegetation, and rising levels in pH of waters in downriver rivers and other bodies of water (Earle and Callaghan 2012). Previously published research shows that biochar, a low-density pyrolyzing material produced by burning biomass under conditions of low temperature and minimal oxygen (Beesley and Marmiroli 2010),” neutralizes the toxic qualities of organic and inorganic pollutants. Biochar may be added to soils with the intention to improve soil functions and to reduce emissions from biomass that would otherwise naturally degrade to greenhouse gases (International Biochar Archive). In order to test this, soil samples were collected at the Joe Johnson, Bonner, Across from Bonner, Brooklyn, and Road Cut mining sites, where biochar had not been introduced to the soil. The mine site has been linked as a key factor to the damages inflicted by acid mine drainage. After the samples were transported to a private research facility, each sample was processed using various metal analysis equipment to provide accurate metal concentration percentages and monitor the effectiveness of Biochar in given percentages corresponding with designated mine sites. Biochar has been proven to retain specific metals (Mendez and Maier 2008) as a means of lowering the direct influence of negative environmental effects thus contributing to the remediation of acid mine drainage sites (Uchimiya, Chang and Klasson 2011). This draws the hypotheses that the biochar will have a high metal retention percentage and allow for sufficient rehabilitation of environments affected by acid mine drainage.
Table 1: The GPS coordinates of the different mine sites that were tested
2. Methods & Materials
Field scientists from Mountain Studies Institute based in Silverton, Colorado, gathered at the Joe John, Brooklyn, Bonner, Across from Bonner, and Road Cut mine sites in Silverton in September of 2011 to collect soil samples. Soil samples were taken back to the facility in order to collect water samples weekly, and take leachate samples at the beginning and the end of experiment was a greenhouse/container experiment where we measured soil water content, soil water pH, and plant growth response. The trial was comprised of concurrent trials where student at Animas High School grew an alpine restoration seed mix in 60 3.5-inch containers under grow lights for 55 days.
2.1 Sample Preparation
When collecting samples, scientists collected one five-gallon bucket-full sample of soil from each of the five mine sites. The soil was then transported to Animas High School, where students prepared the samples in containers. Students first sieved the sample in order to separate out the soil from the rocks, as well as to break the particles down to less than 2 mm fractions. 200 ml of each soil sample were weighed in grams and recorded on the data sheet. Once the samples had been broken down, a mix of seeds was separated into units of one gram, in addition to 4 Lupine seeds. The seed mixture was added in a labeled container to the soil sample later on in the experiment. After the soil and seed mixtures had been sorted out, the biochar volumes were measured in accordance to the equalities: 0 ml=0%, 20ml=10%, 40ml=20%, and 60ml=30% of biochar per 200 ml soil sample. These volumes were then converted and recorded as mass measured in grams. Once all of the data was recorded, the soil and biochar were thoroughly mixed together in their respective containers. The samples were mixed together by combining the two in a mixing container as completely as possible, and then returning the samples to their labeled containers. Next, the mixture was saturated with water and left to drain overnight. Once drained, the biochar and soil were gently mixed back together, and the seed combination was added to the mixture. Once the seeds were added, the mixture was saturated, drained, and finally weighed. After the mass had been recorded, the sample was covered and left to germinate for 72 hours at a warm temperature. Once the seeds had germinated, the samples were watered once a week with 100 ml of tap water. The pH of the tap water was recorded before irrigation.
2.2 Measurements Taken Throughout Trial
After the samples had been correctly set up, measurements were taken once a week for the duration of the study. Students first documented the samples photographically, noting the site plot, treatment, date, and team on a dry erase board that was included in the photo. Students then recorded the average height (rounded to the nearest 5 mm) of the green vegetation that had grown in the sample. Students then irrigated the samples with 100 ml of tap water, and recorded the mass of the sample in grams. Mass of the sample was also taken prior to irrigation. Once the water had been drained, the pH of the leachate was recorded. For the first and last irrigations of the study, the leachate was collected for metals analysis.
2.3 Metals Analysis
In order to collect the leachate, the initial leachate samples were filtered with filter paper into 60 ml plastic bottles. Upon collection, they were refrigerated, in accordance with standard protocol. They were then fixed with 12 drops of 70% nitric acid by mass in order to keep the dissolved metals in their dissolved forms. Once the leachate had been collected, the samples were sent to a lab in Fort Collins, Colorado for metals analysis in an ICP-MS instrument. ICP-MS stands for Inductively Coupled Plasma Mass Spectrometry. The first step in the process is converting the sample to an aerosol in order to excite the particles and carry them into the plasma. The objective of the plasma is to strip the electrons from the analyte atoms and to generate ions. The ions are then transmitted through a spectrometer and filtered by their mass, thereby emitting a mass spectrum in correlation to the element of the particles. The elements are identified by associating the peak in the spectrum to the known isotope concentration of the element. (Thermo Fisher, 12-14).
3. Results
The results of distributing varying concentrations of biochar throughout different mine sites in Silverton, Colorado, suggest that biochar successfully retains harmful metals in solution, such as aluminum, cadmium, lead, copper, zinc, iron, arsenic, manganese, and selenium to a certain degree. Although some data remains inconsistent in terms of metal retention for some concentrations of biochar, specifically concentrations of biochar at 20% and 30%, the overall spectrum of the data shows that biochar can cause a significant reduction of metal concentration in soil.
Lead displayed a significant decrease in concentrations when exposed to 10% biochar concentration. After being exposed to higher concentrations, however, the lead concentrations began to increase at a rate that was slower than the initial decrease, but still consistent. aluminum showed a significant reduction in concentration when exposed to a 10% biochar concentration.
Iron presented high levels in the leachate, but once introduced to biochar, those levels started decreasing gradually. Once the biochar concentrations reached above 20%, however, levels of iron started spiking.
Because of the significant differences between biochar concentrations and their effectiveness in retaining both aluminum and iron, there is most likely no relationship between biochar concentrations and their abilities to retain iron or aluminum. Once the biochar was mixed with a 20% concentration of biochar, however, there was a slight decrease in levels of copper present in the leachate.
Manganese did not follow this trend. Instead of the amounts of metals decreasing after being introduced to biochar, the levels increased until they were exposed to 20% biochar concentrations, when the levels at last started to decrease.
The levels of Cadmium were so low that there was hardly any change when the sample was introduced to biochar. Nevertheless, levels of cadmium did slightly decrease after being mixed with concentrations of biochar.
Zinc simply showed erratic results, making it difficult to draw any substantial data from the results.
The average retention rate for all metals at 10% biochar concentration was 32.7%, 20% biochar concentration was 22.09%, and 30% biochar concentration averaging out to 35.9%. The median retention rates of all of these biochar concentrations was 30.23%, with the mode being 52%. The range between all of rates of retention represented a difference of 208, with levels ranging between -113% retention and 95%.
Figure 2: the overall retention rate of metals at the given percent biochar
4. Discussion
Because there were several obvious retentions in certain metal levels, there is a possibility that biochar is only effective in retaining certain metals at a certain concentration. For example, it is apparent in the graphs that biochar is somewhat successful in retaining aluminum and lead levels at concentrations equal to or less than 20%, because both metal values started spiking after 20% biochar concentrations. Iron levels are also reduced when exposed to anything less than a 20% biochar concentration. Contrary to these results, both manganese and copper started decreasing in values only when exposed to concentrations greater than 20%. Regardless of the biochar concentrations zinc was exposed to, there was no apparent reaction to the added substance, suggesting biochar does not help retain zinc in solution. Because of the variance in results, we have concluded that there is no specific biochar concentration that will be successful in retaining the harmful metals listed above. There is a slight possibility, however, that the inclusion of biochar in the soil sample raised the pH of the metals, thereby decreasing the potential of the metals in solution to sorb to the biochar, which is why we think that our results were so varied.
Because of the variance in our results, we cannot prove our hypothesis valid, as there was no uniform decrease in the metal values. This variance may be related to errors in measuring metal values, as there were some values that seemed very random or odd in relation to other measurements in the same study. These errors would have affected the results in a random way, either increasing or decreasing the estimated retention rate, as was obvious when the calculated average retention rate was greater than negative or greater than 100%. It is also not certain that the plants all got the same treatment, or the leachate was collected the same, which would result in randomized data once again. Because this is an original study in the respect that no other studies have tested varying concentrations of biochar, our results are not comparable to another published study. One study, A review of Biochars’ Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soils, did state in their abstract that “Specific mechanisms of contaminant-biochar retention and release over time and the environmental impact of biochar amendments on soil organisms remain somewhat unclear but must be investigated to ensure that the management of environmental pollution coincides with ecological sustainability,” stressing the importance of biochar as a combatant towards certain types of environmental pollution, including acid mine drainage. For future studies, it may prove beneficial to send the soil samples to two different metals analysis centers, in order to compare results and confirm the accuracy of the values.
5. Acknowledgements
The authors of this paper are grateful to Mountain Studies Institute and the River Watch Program for their assistance with sample preparation and statistical analysis.
6. References
Beesley, Luke, Eduardo Moreno-Jimenéz, Jose L. Gomez-Eyles, Eva Harris, Brett Robinson, and Tom Sizmur. A Review of Biochars ’ Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soils. Publication. A Review of Biochars' Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soilsand Restoration of Contaminated Soils (Tom Sizmur). El Sevier, 2011. Web. 15 Apr. 2012.
Beesley, L. and Marmiroli, M., 2010. “The immobilization and retention of soluble arsenic, cadmium and zinc by biochar.” Environmental Pollution pp. 1
Colorado Department of Public Health and Environment 2009. System Classifacation and Water Quality Standards.”Water Quality Regulations AnimasPDF
Colorado Department of Public Health and Environment 2009.“The Basic Standards for Ground Water.” The Water Quality Control CommissionPDF
Earle, J., Callaghan, T., “Impacts of mine drainage on aquatic life, water uses, and man-made structures.” in Coal Mine Drainage Prediction and Prevention Department of Environmental Protection, Harrisburg, PA 17105 pp. 4-1 - 4-10
“From First Principles: An Introduction to the ICP-MS Technique.” Thermo Fisher Scientific. Web. 18 Apr 2012.http://www.thermo.com/CMA/PDFs/Various/File_2512.pdf
Jonesw, DL., Rousk, J., Edwards-Jones, G., DeLuca,T.H., Murphy, D.V.Murphy, 2011 “Biochar-mediated changes in soil quality and plant growth in a three year trial.” Soil Biology and Biochemistry pp. 113-124
McGuinness, S., 1999. Treatment of acid mine drainage. RESEARCH PAPER 99/10 House of Commons pp. 1-5
Mendez, M. and Maier, R., 2008. “Phytostabilization of Mine Tailings in Arid and Semiarid Environments- An Emerging Remediation Technology.” Department of Soil, Water, and Environmental Science, University of Arizona, Tucson, Arizona, USA pp.278-280
Uchimiya, M., Chang,, S., Klasson, T., 2011“Screening biochars for heavy metal retention in soil: Role of oxygen functional groups” Journal of Hazardous Materials, Volume 190, Issues 1–3, 15 June 2011, pp. 432-441
"What Is Biochar?" International Biochar Initiative. 2012. Web. 02 May 2012. <http://www.biochar-international.org/biochar>.